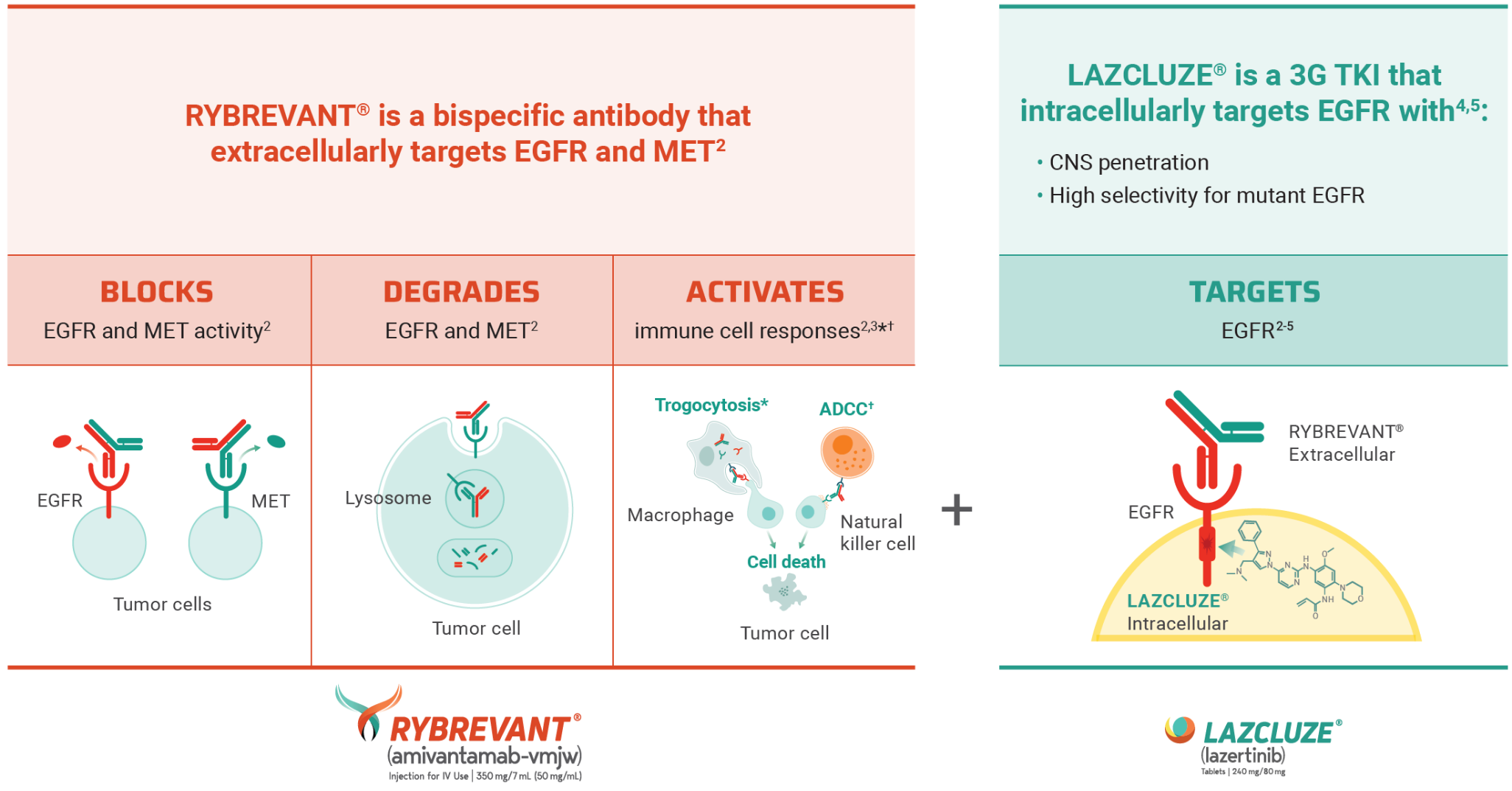
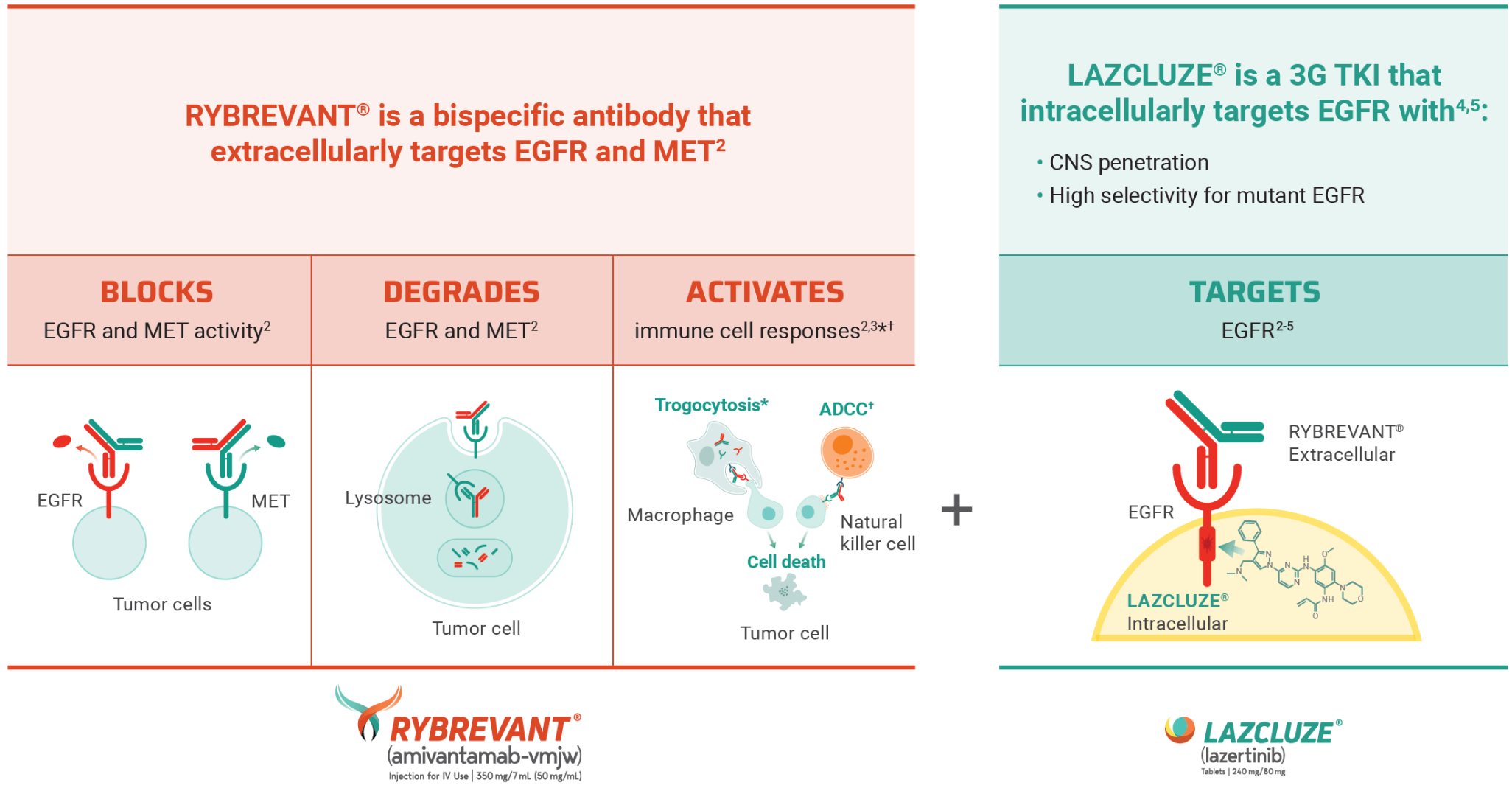
MARIPOSA Trial: A Brief Overview
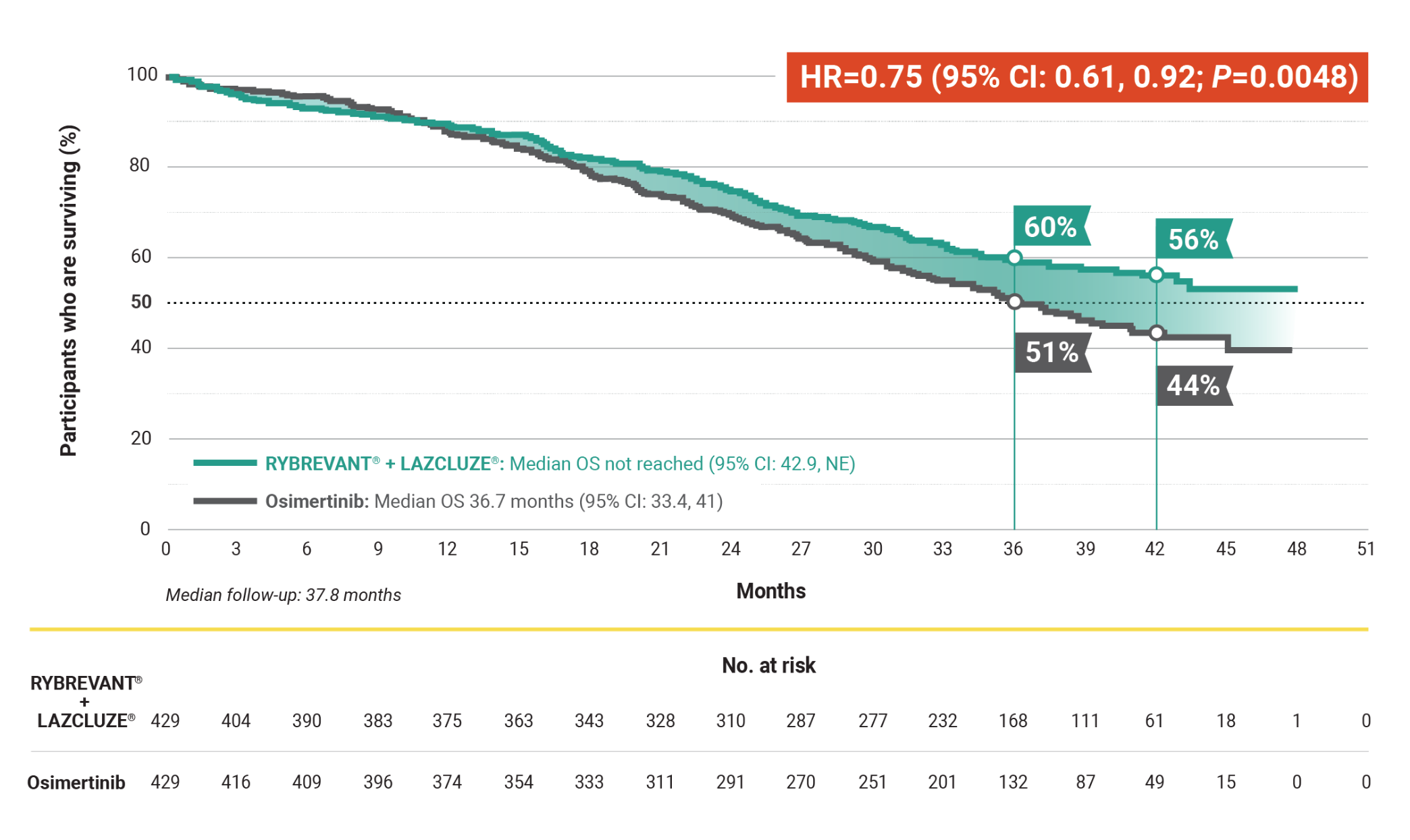
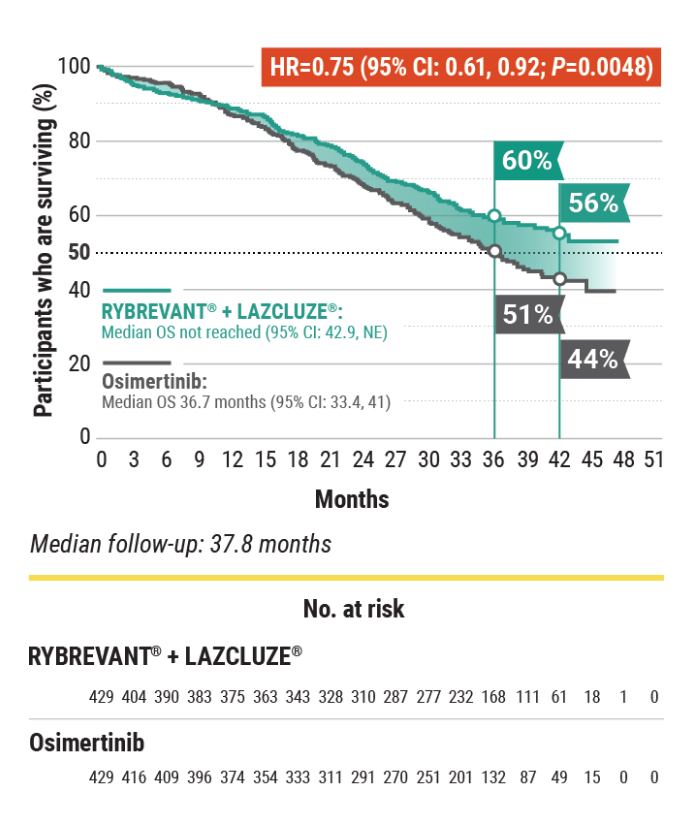
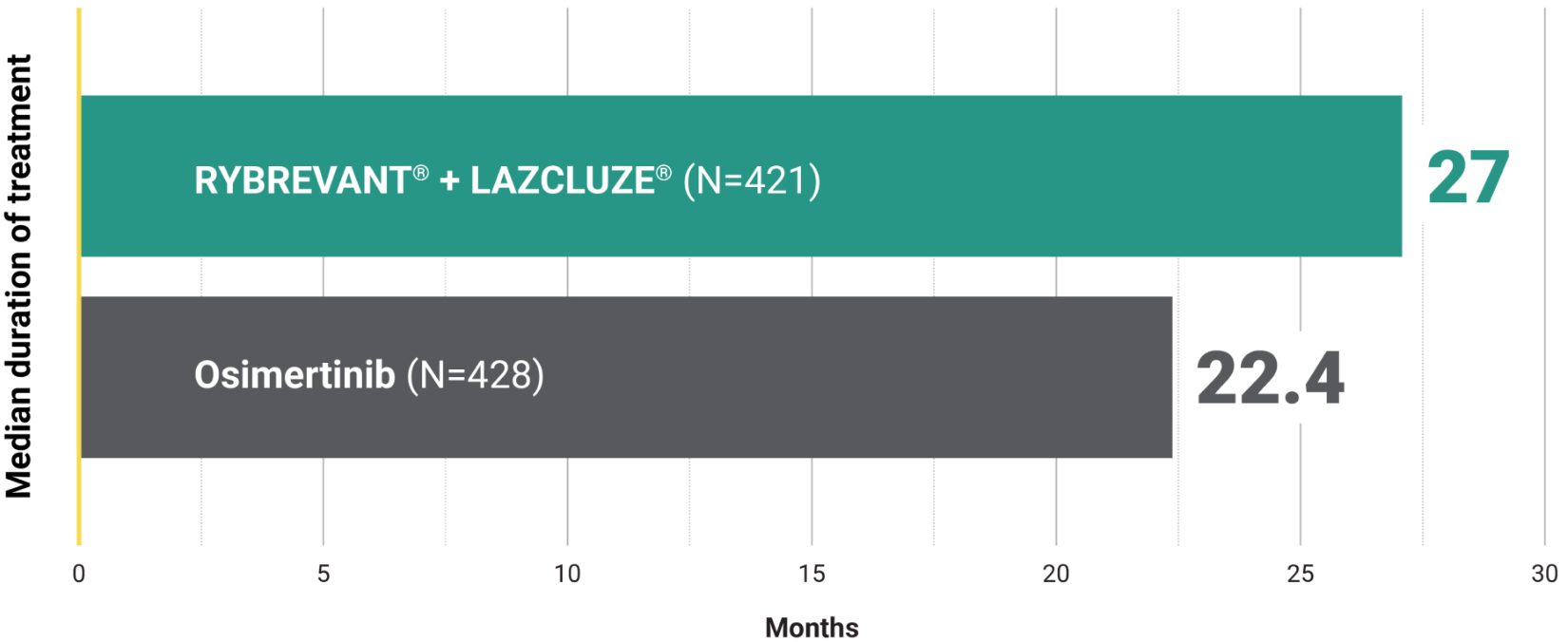
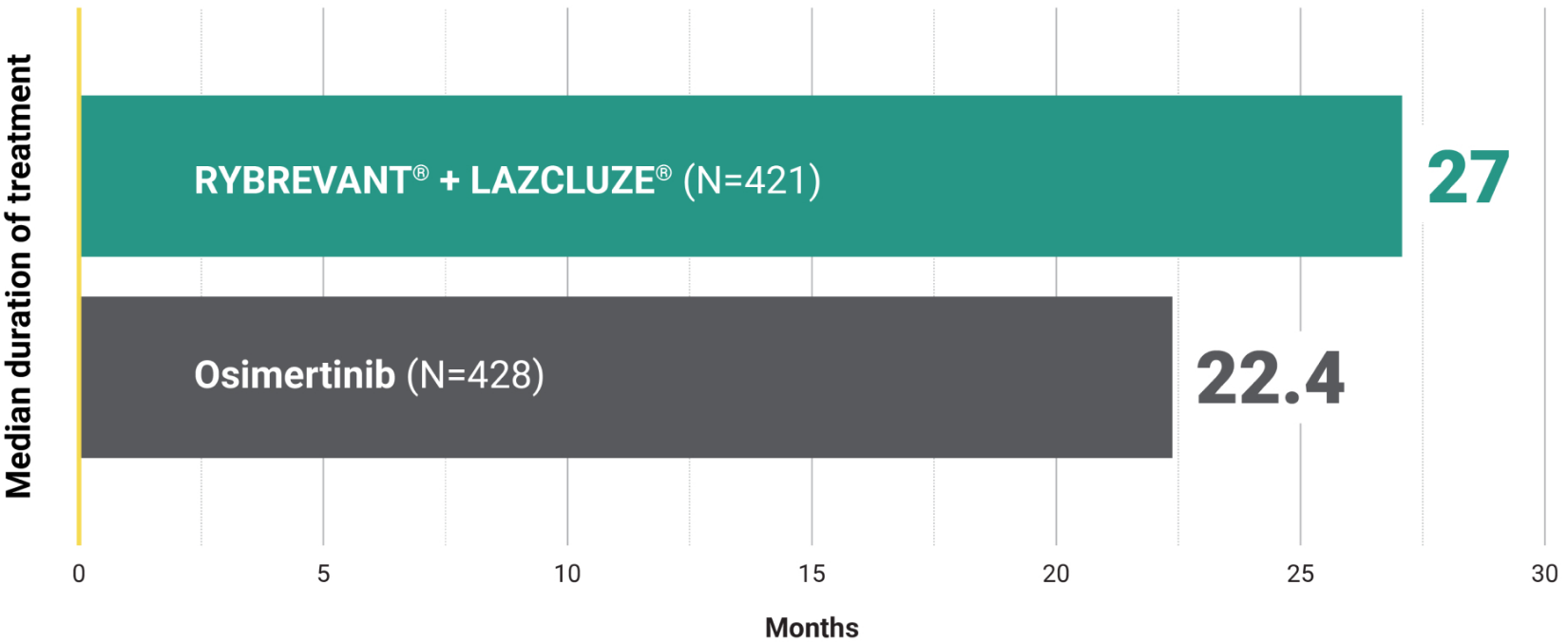
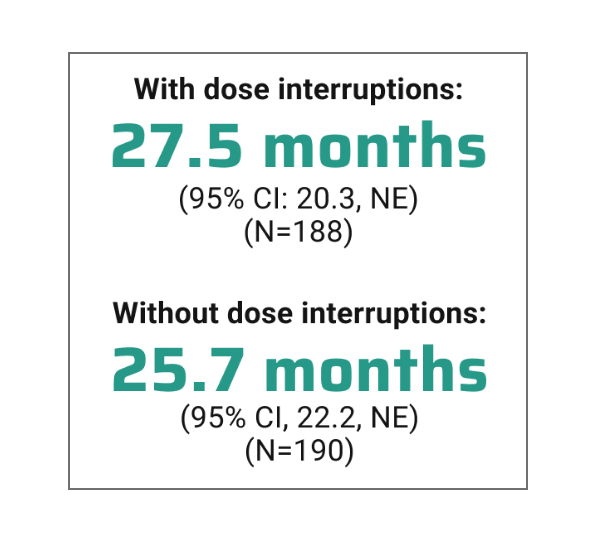
Overall Survival – Secondary Endpoint
For first-line treatment of adult patients with locally advanced or metastatic EGFR+ NSCLC
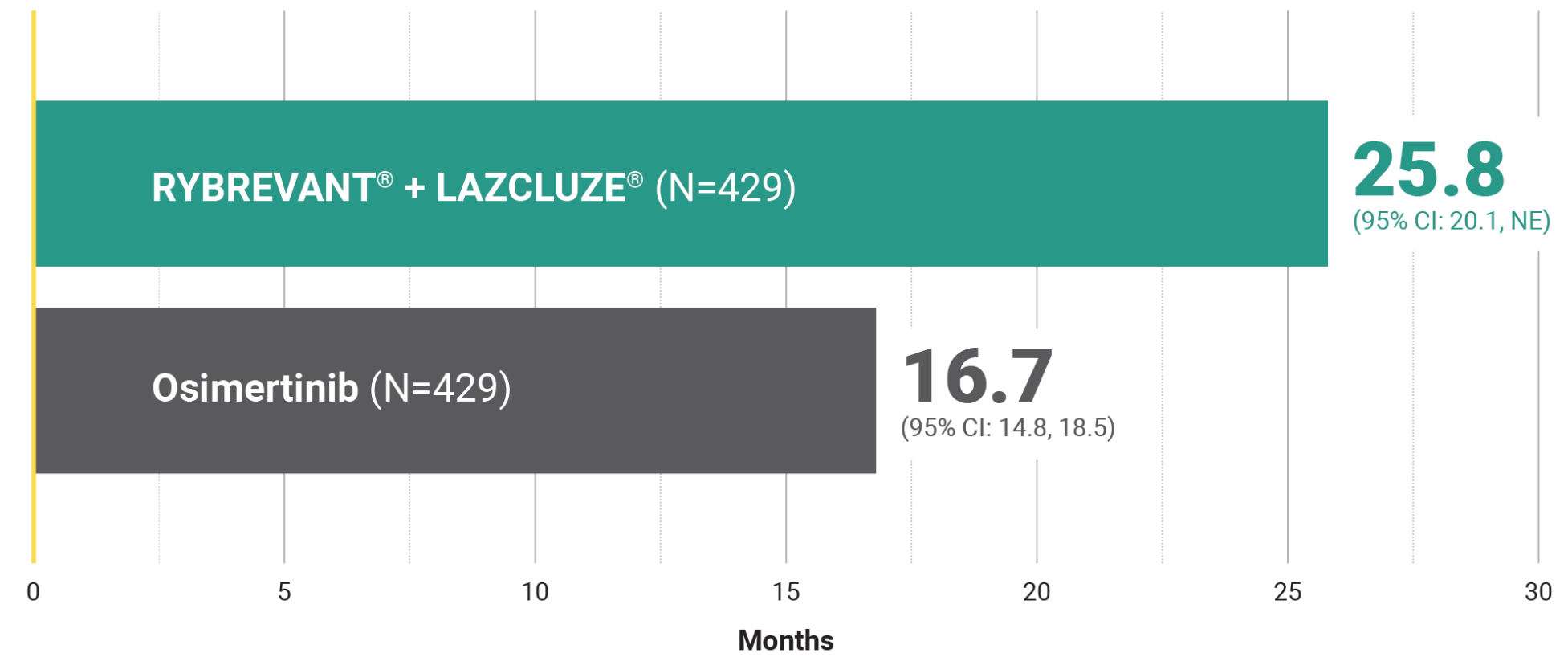
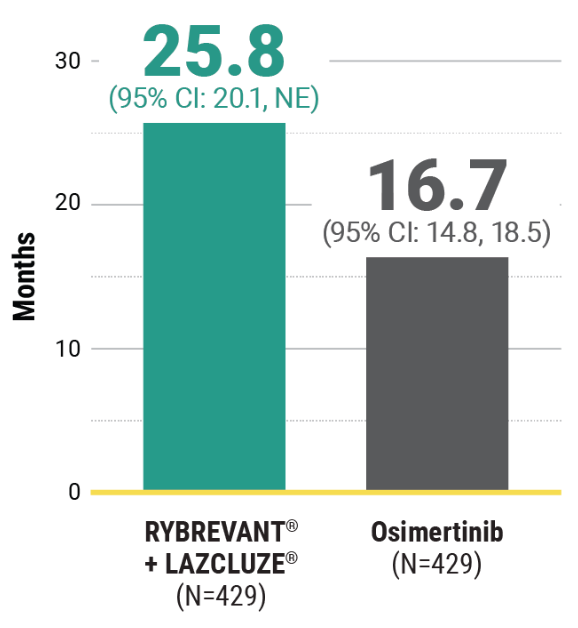
Unmatched survival: RYBREVANT® + LAZCLUZE® delivers superior overall survival vs osimertinib with proven durability1,2
Median overall survival not reached with RYBREVANT® + LAZCLUZE® and projected to exceed 4 years3
Based on modeling using observed HR and mOS in the osimertinib group, assuming exponential distribution of OS in both arms. The baseline factors were: mutation type, race, brain metastases, age, sex, ECOG PS, and weight. This is a conservative estimate; final results may vary.3
CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance status; EGFR, epidermal growth factor receptor; HR, hazard ratio; mNSCLC, metastatic non–small cell lung cancer; mOS, median overall survival; NE, not estimable; NSCLC, non–small cell lung cancer; OS, overall survival.
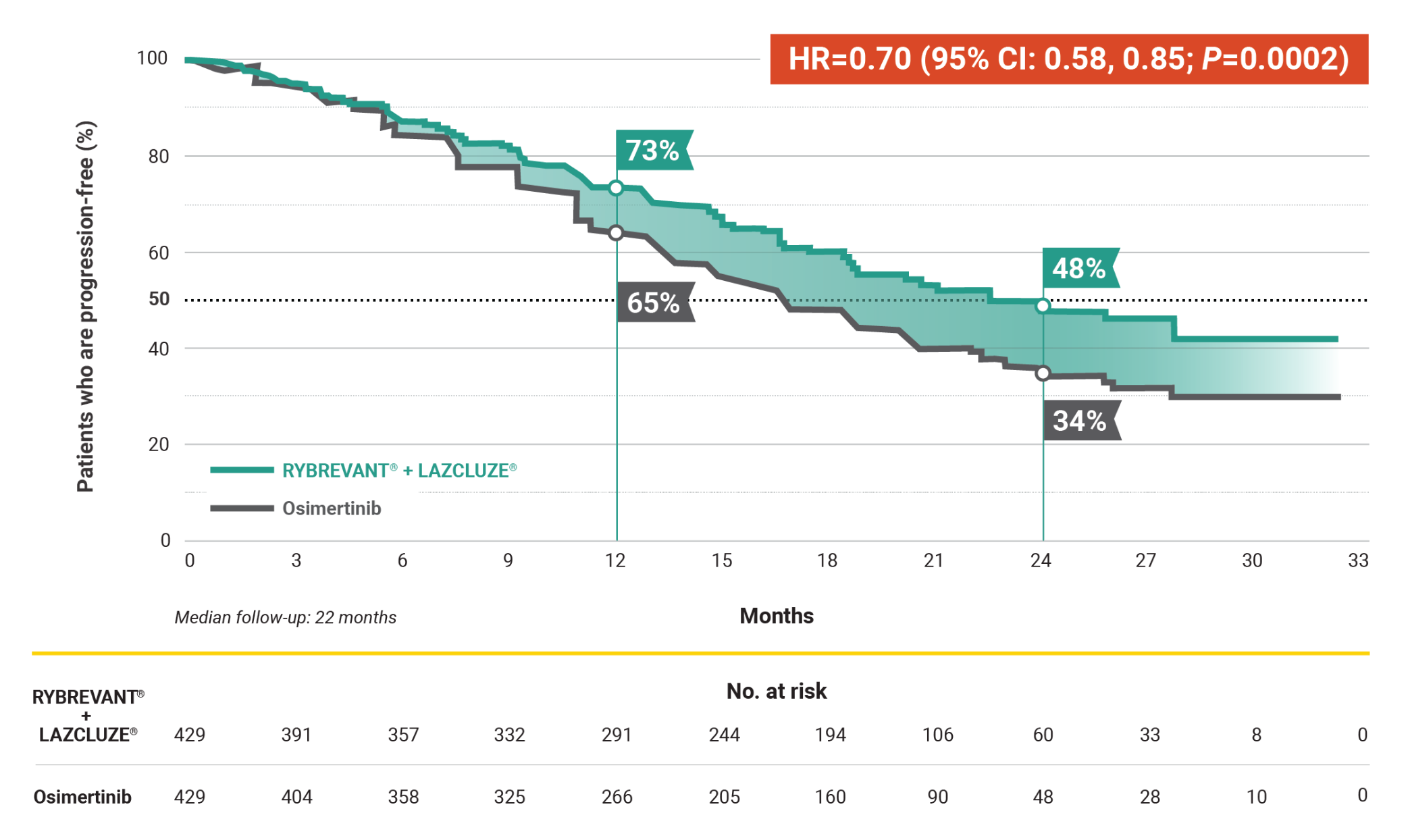
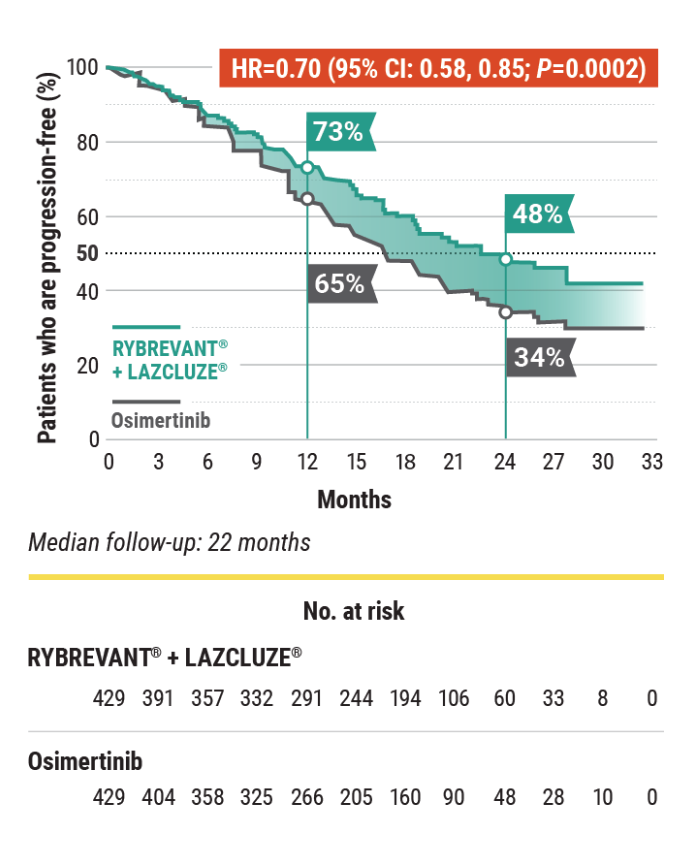
Progression-Free Survival – Primary Endpoint
For first-line treatment of adult patients with locally advanced or metastatic EGFR+ NSCLC
Superior PFS vs osimertinib with a chemo-free combination1,4
RYBREVANT® + LAZCLUZE® demonstrated a statistically significant reduction in the risk of progression or death by 30% vs osimertinib1,5
7.1-month improvement in mPFS vs osimertinib1
- 23.7-month (95% CI: 19.1, 27.7) mPFS with RYBREVANT® + LAZCLUZE® vs 16.6-month (95% CI: 14.8, 18.5) mPFS with osimertinib
In the non-registrational LAZCLUZE® arm, mPFS was 18.5 months (95% CI: 14.8, 20.1)5
National Comprehensive Cancer Network® (NCCN®)
PREFERRED FIRST-LINE THERAPY6*
First-line amivantamab-vmjw (RYBREVANT®) + lazertinib (LAZCLUZE®) is the only NCCN Category 1
preferred multitargeted treatment option for patients with EGFR+† mNSCLC.‡§
EGFR mutation discovered prior to first-line systemic therapy.6
EGFR exon 19 deletion or exon 21 L858R mutations.6
See the current NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for detailed recommendations, including other treatment options.
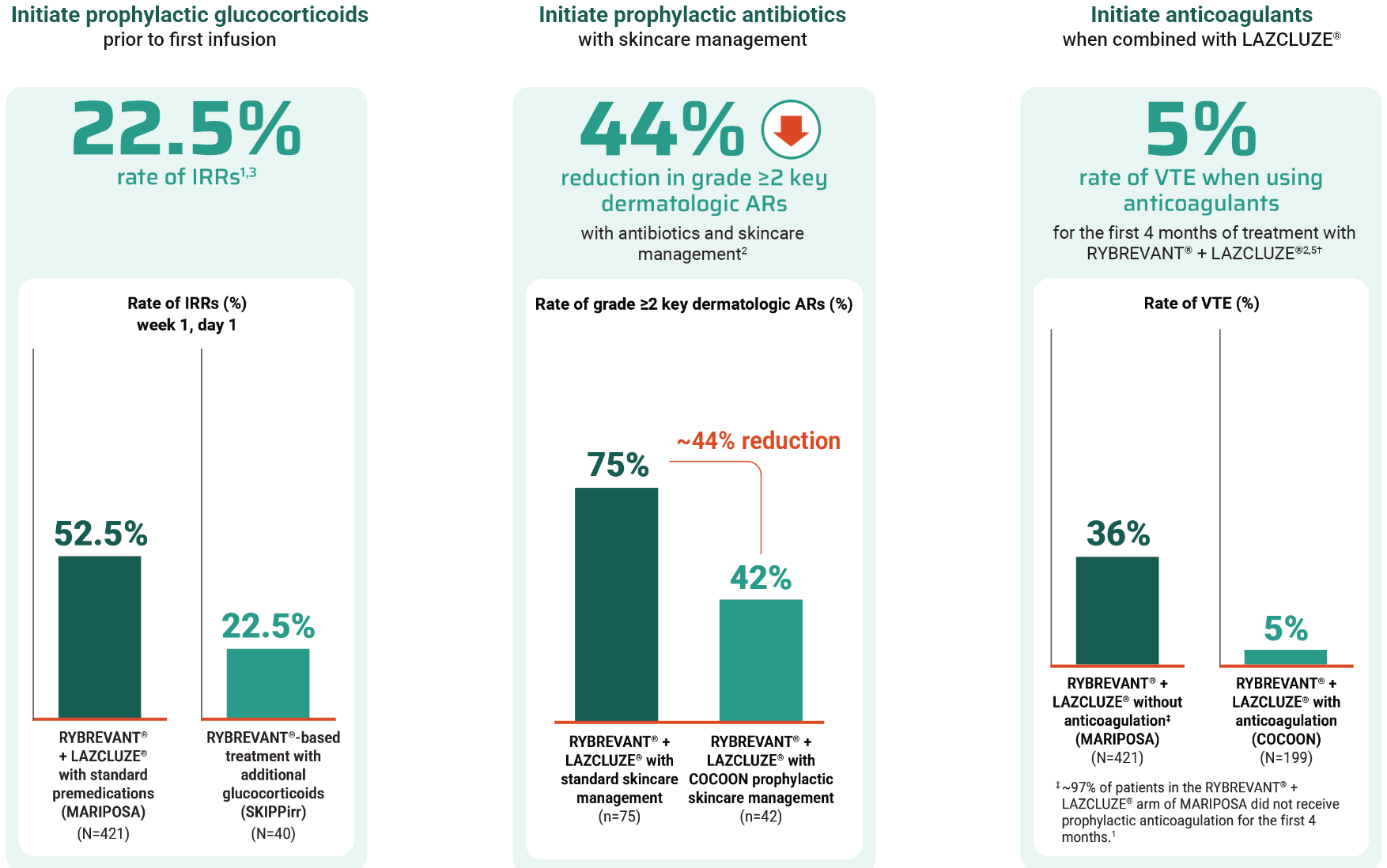
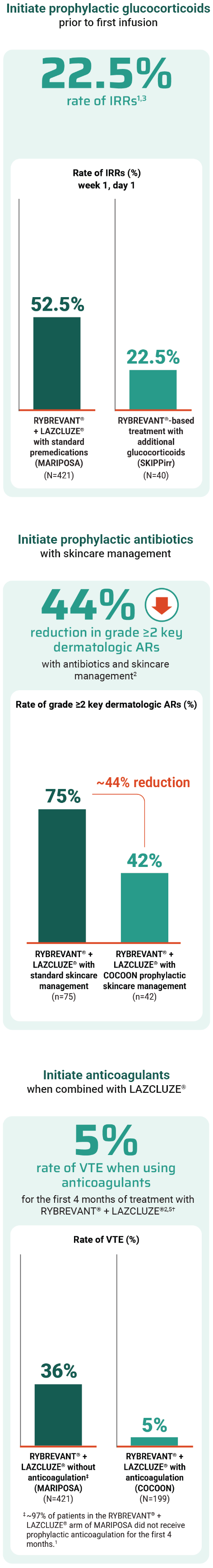
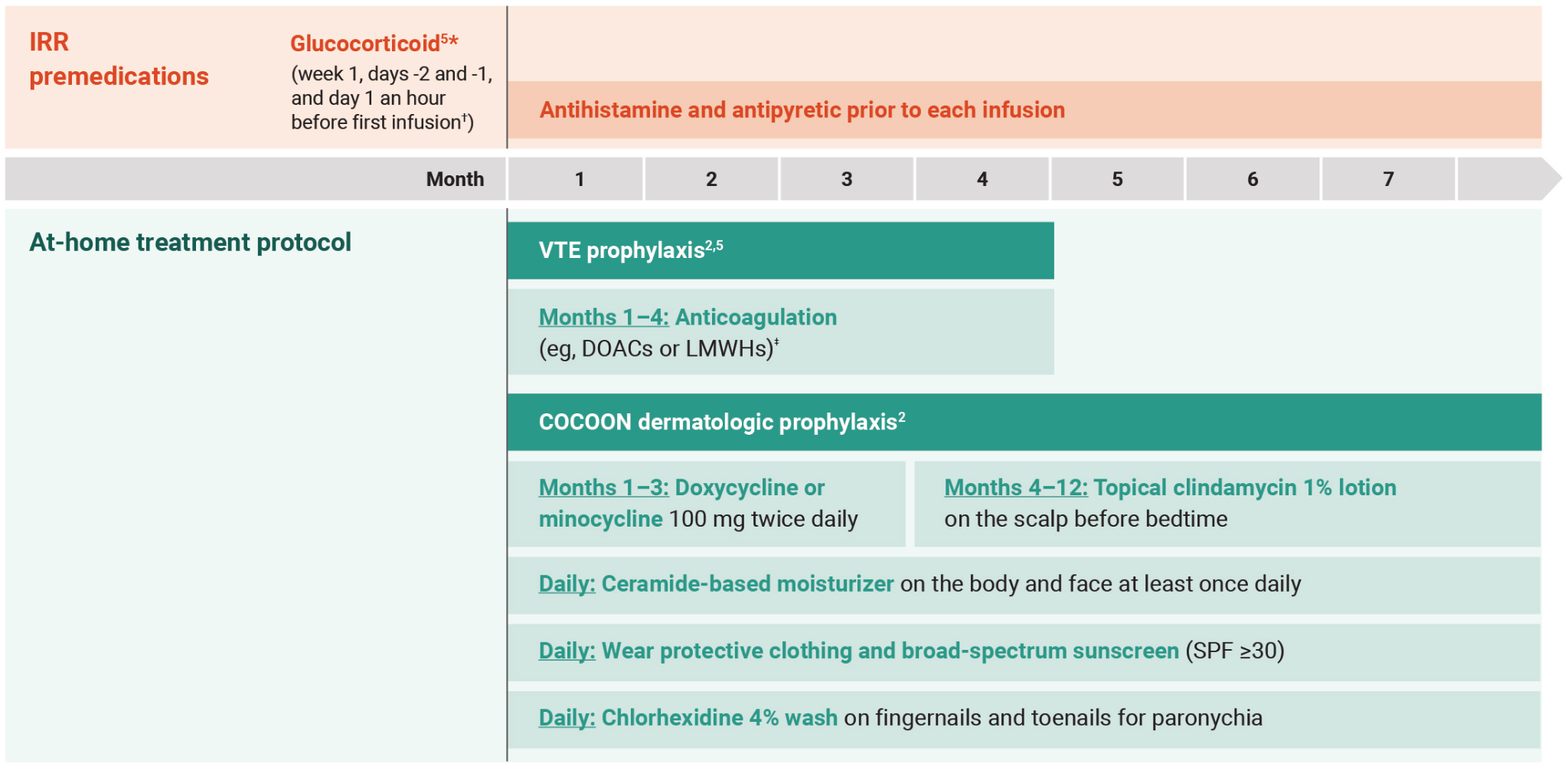
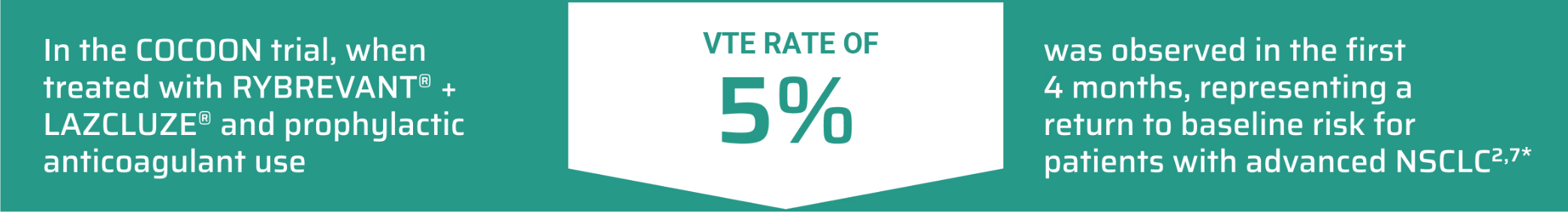
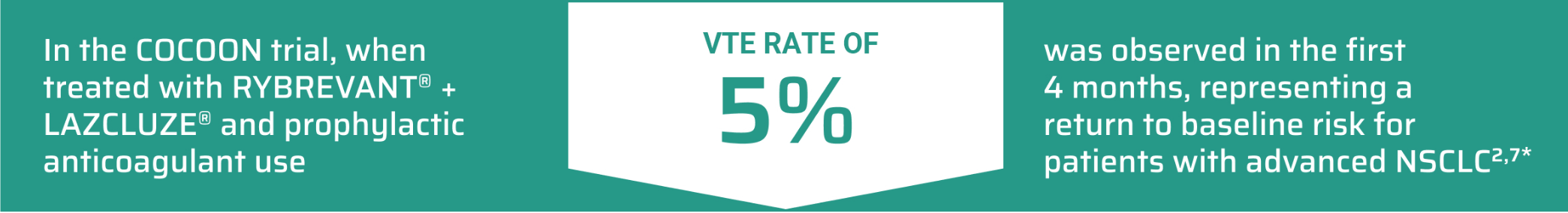
Prophylactic anticoagulation is recommended at the time of initiation to prevent venous thromboembolic events.6
EGFR, epidermal growth factor receptor; mNSCLC, metastatic non–small cell lung cancer; mPFS, median progression-free survival; NCCN, National Comprehensive Cancer Network; PFS, progression-free survival.
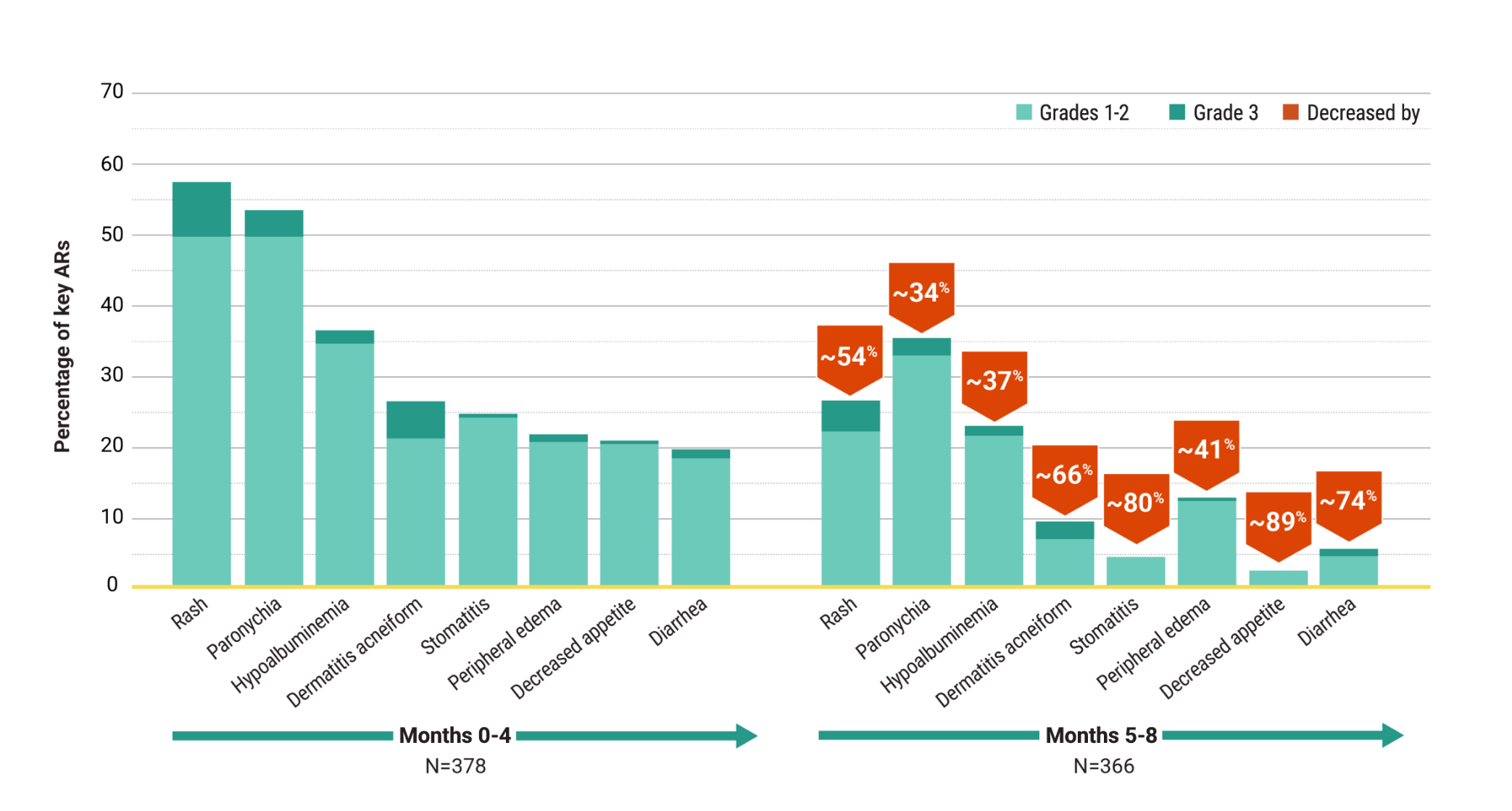
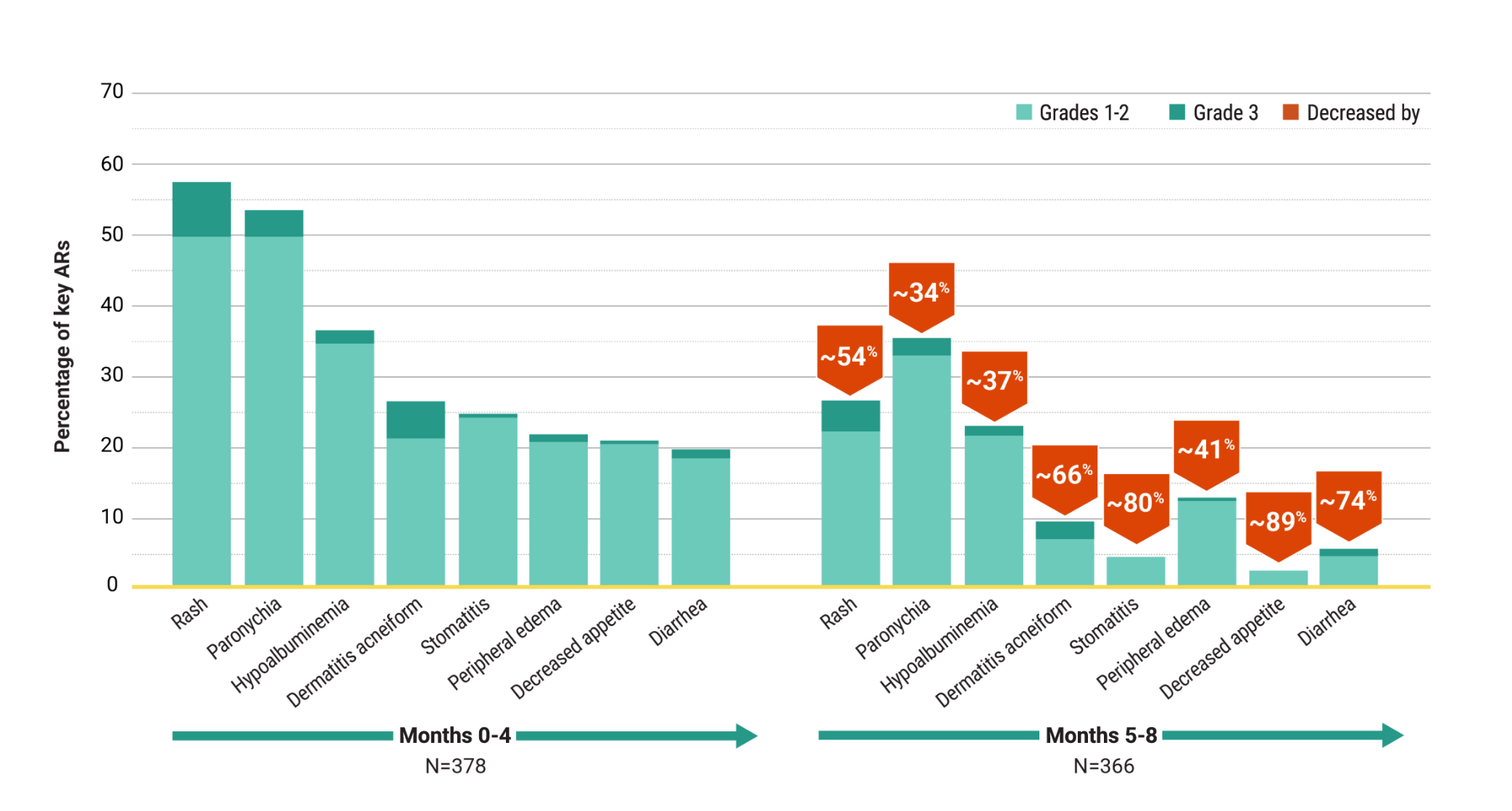
Other Endpoints
For first-line treatment of adult patients with locally advanced or metastatic EGFR+ NSCLC
High and durable responses with a chemo-free combination1,4
Overall response rate1
- ORR was 78% (95% CI: 74, 82) with RYBREVANT® + LAZCLUZE® (N=429) and 73% (95% CI: 69, 78) with osimertinib (N=429)
- 73% of patients treated with RYBREVANT® + LAZCLUZE® achieved a PR and 70% of patients treated with osimertinib
- 5.4% of patients treated with RYBREVANT® + LAZCLUZE® achieved a CR and 3.5% of patients treated with osimertinib
Median duration of response1
This was a prespecified analysis and was not powered to show statistical significance.
~1.5X mDOR with RYBREVANT® + LAZCLUZE®1
CR, complete response; mDOR, median duration of response; ORR, overall response rate; PR, partial response.
CNS Data
The National Comprehensive Cancer Network® (NCCN®) recommends
Amivantamab-vmjw (RYBREVANT®)-based regimens*: The only NCCN preferred combination options for brain metastases in patients with EGFR+† mNSCLC1,7‡
Intracranial PFS§ at 36 months in subjects with intracranial lesions at baseline3
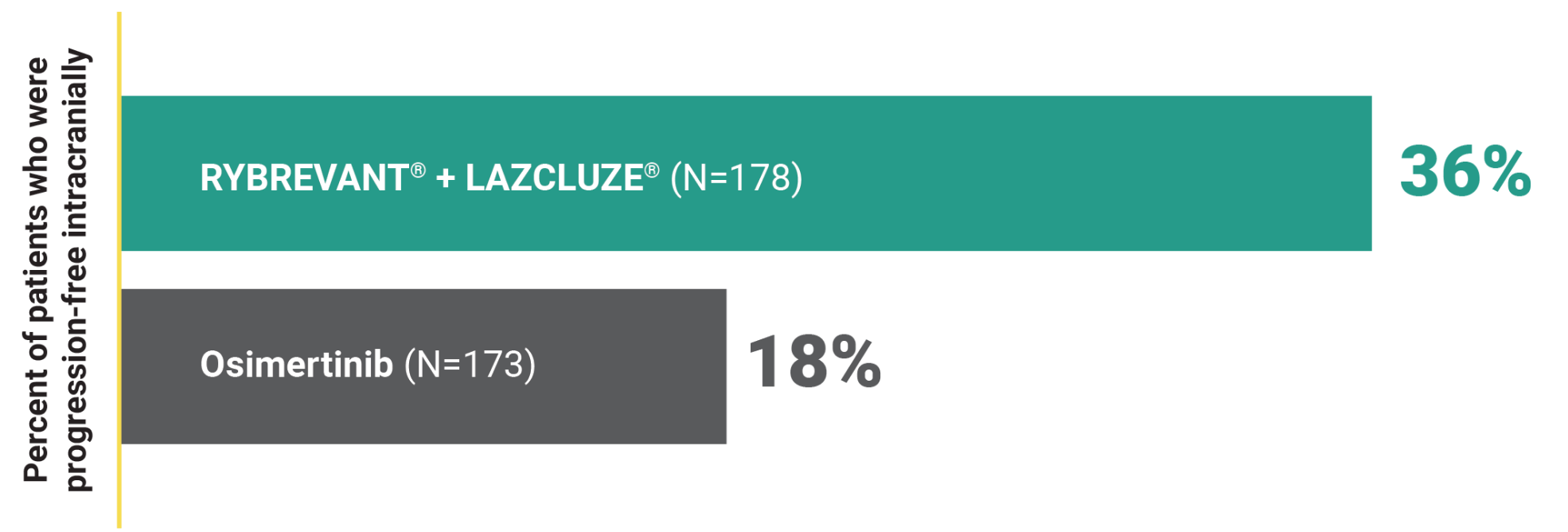
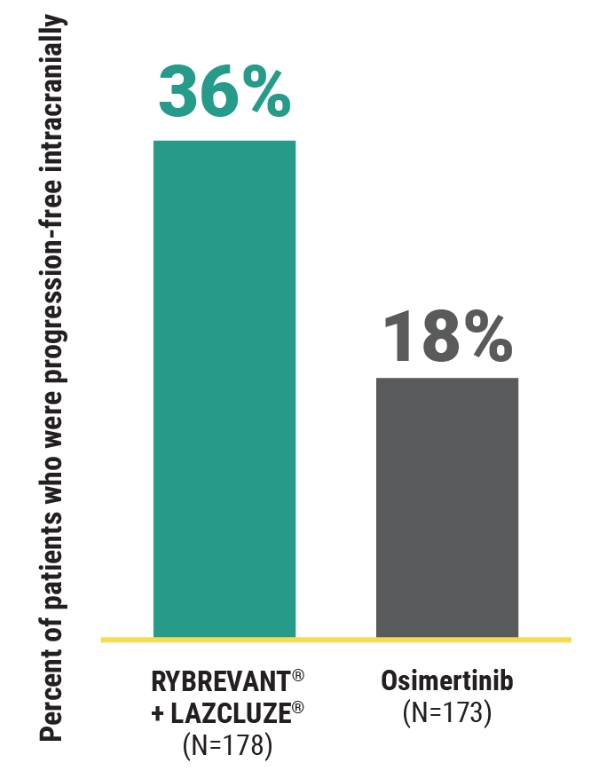
This was a prespecified secondary analysis and was not powered to show statistical significance.
2X intracranial PFS at 36 months with RYBREVANT® + LAZCLUZE®3
Amivantamab-vmjw + lazertinib; amivantamab-vmjw + carboplatin + pemetrexed.
EGFR exon 19 deletion or exon 21 L858R mutations.7
See the current NCCN Guidelines® for detailed recommendations, including other treatment options.
Based on median follow-up of 37.8 months.3
For first-line treatment of adult patients with locally advanced or metastatic EGFR+ NSCLC
High intracranial ORR with durable intracranial DOR3,8
Intracranial ORR in subjects with intracranial lesions at baseline*
- Intracranial ORR was 78% (95% CI: 71, 84) with RYBREVANT® + LAZCLUZE® (N=180) and
77% (95% CI: 71, 83) with osimertinib (N=186)3
- Intracranial CR was 64% with RYBREVANT® + LAZCLUZE® and 59% with osimertinib8
- Intracranial PR was 14% with RYBREVANT® + LAZCLUZE® and 19% with osimertinib8
This was a prespecified exploratory analysis and was not powered to show statistical significance.
Intracranial DOR in confirmed responders with intracranial lesions at baseline8*†
Median intracranial duration of response
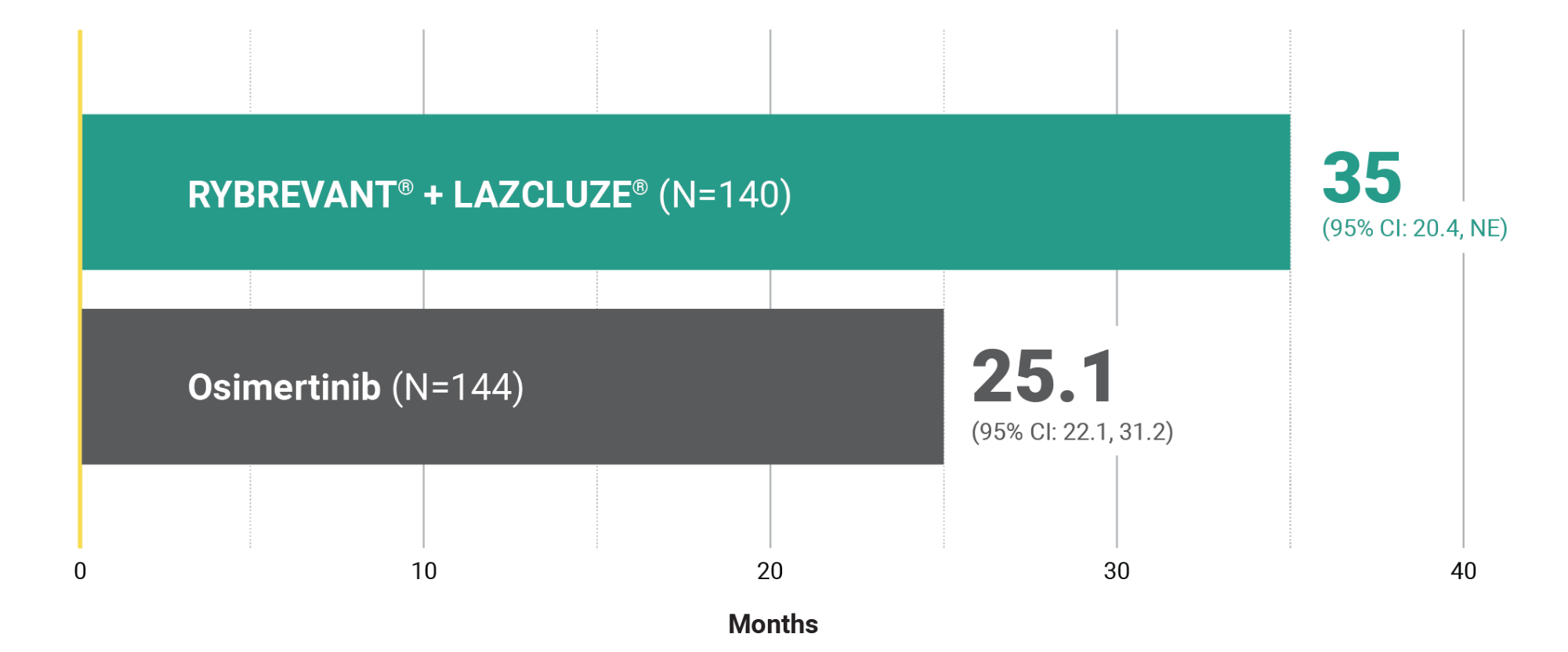
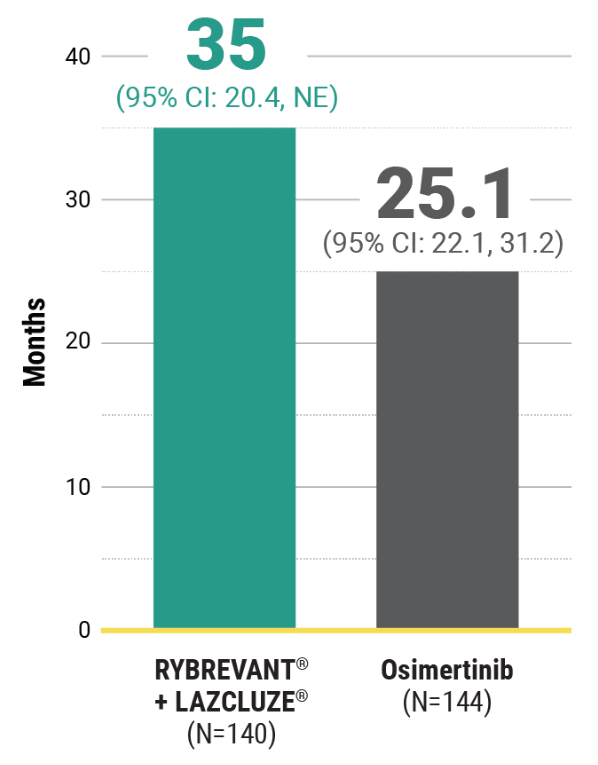
This was a prespecified exploratory analysis and was not powered to show statistical significance.
Based on median follow-up of 37.8 months.3
Based on subgroup of subjects with history of brain metastasis. CR and PR do not have to be confirmed.8
~1.5X intracranial DOR with RYBREVANT® + LAZCLUZE®8
1L, first-line; CNS, central nervous system; DOR, duration of response; icDOR, intracranial DOR.
NCCN PREFERRED THERAPY FOR BRAIN METASTASES1,7
Amivantamab-vmjw (RYBREVANT®)-based regimens* including amivantamab-vmjw (RYBREVANT®) + lazertinib (LAZCLUZE®) are the only NCCN preferred combination treatment options for brain metastases in patients with EGFR+† mNSCLC.‡
Amivantamab-vmjw + lazertinib; amivantamab-vmjw + carboplatin + pemetrexed.
EGFR exon 19 deletion or exon 21 L858R mutations.7
See the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for detailed recommendations, including other treatment options.
Study Design
For first-line treatment of adult patients with locally advanced or metastatic EGFR+ NSCLC
MARIPOSA: Evaluating the first and only multitargeted combination in first-line EGFR+ mNSCLC vs osimertinib1,4
MARIPOSA was an active-controlled, multicenter, phase 3 trial. Patients with asymptomatic or previously treated and stable intracranial metastases were eligible to enroll. Patients received treatment until disease progression or unacceptable toxicity. The evaluation of efficacy relied upon comparison between RYBREVANT® in combination with LAZCLUZE®, and osimertinib.1,9
The largest phase 3 trial and the only one that required serial brain MRIs for all patients, providing accurate detection of CNS progression in patients with 1L EGFR+ disease5,9-20*
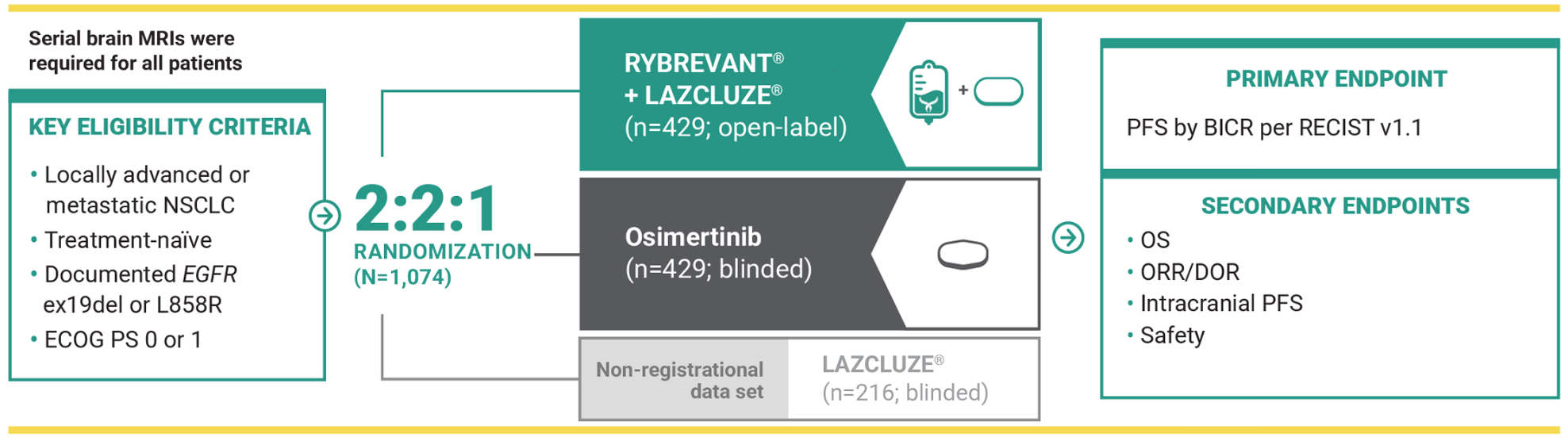
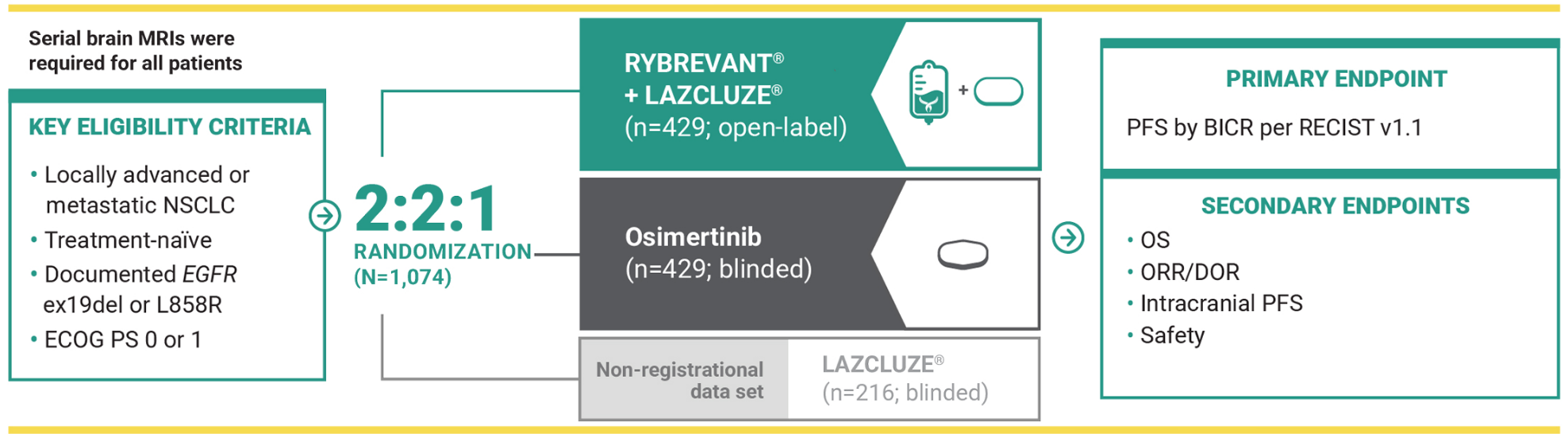
LAZCLUZE® monotherapy arm was included to assess the contribution of the components.5
MARIPOSA was the largest phase 3 trial that evaluated 1L treatment in patients with EGFR+ mNSCLC as of April 2025.5,9-20
Serial brain MRIs were conducted for all patients to assess intracranial progression and response5
- Serial brain MRIs were performed at baseline and either every 8 weeks for the first 30 months and 12 weeks thereafter (for patients with a history of brain metastases) or every 24 weeks (for patients without a history)
BICR, blinded independent central review; ex19del, exon 19 deletion; MRI, magnetic resonance imaging; RECIST, Response Evaluation Criteria in Solid Tumors.
See more of the clinical study results
References:
1. RYBREVANT® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
2. Yang JC-H, Kim YJ, Lee S-H, et al. Amivantamab plus lazertinib vs osimertinib in first-line EGFR-mutant advanced NSCLC: Final overall survival from MARIPOSA. Presented at: European Lung Cancer Congress; March 26-29, 2025; Paris, France.
3. Yang JC-H, Lu S, Hayashi H, et al; for the MARIPOSA Investigators. Overall survival with amivantamab-lazertinib in EGFR-mutated advanced NSCLC. N Engl J Med. Published online September 7, 2025. doi:10.1056/NEJMoa2503001
4. LAZCLUZE® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
5. Cho BC, Lu S, Felip E, et al; MARIPOSA Investigators. Amivantamab plus lazertinib in previously untreated EGFR-mutated advanced NSCLC. N Engl J Med. 2024;391(16):1486-1498. doi:10.1056/NEJMoa2403614
6. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Non-Small Cell Lung Cancer. V.3.2026. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed December 26, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
7. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Central Nervous System Cancers V.3.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed December 18, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
8. Data on file. Janssen Biotech, Inc.
9. Cho BC, Lu S, Felip E, et al; MARIPOSA Investigators. Amivantamab plus lazertinib in previously untreated EGFR-mutated advanced NSCLC. Supplementary Appendix. N Engl J Med. 2024;391(16):1486-1498. doi:10.1056/NEJMoa2403614
10. Yang JCH, Wu YL, Schuler M, et al. Afatinib versus cisplatin-based chemotherapy for EGFR mutation-positive lung adenocarcinoma (LUX-Lung 3 and LUX-Lung 6): analysis of overall survival data from two randomised, phase 3 trials. Lancet Oncol. 2015;16(2):141-151. doi:10.1016/S1470-2045(14)71173-8
11. Wu YL, Cheng Y, Zhou X, et al. Dacomitinib versus gefitinib as first-line treatment for patients with EGFR-mutation-positive non-small-cell lung cancer (ARCHER 1050): a randomised, open-label, phase 3 trial. Lancet Oncol. 2017;18(11):1454-1466. doi:10.1016/S1470-2045(17)30608-3
12. Rosell R, Carcereny E, Gervais R, et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2012;13(3):239-246. doi:10.1016/S1470-2045(11)70393-X
13. Nakagawa K, Garon EB, Seto T, et al. Ramucirumab plus erlotinib in patients with untreated, EGFR-mutated, advanced non-small-cell lung cancer (RELAY): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20(12):1655-1669. doi:10.1016/S1470-2045(19)30634-5
14. Kawashima Y, Fukuhara T, Saito H, et al. Bevacizumab plus erlotinib versus erlotinib alone in Japanese patients with advanced, metastatic, EGFR-mutant non-small-cell lung cancer (NEJ026): overall survival analysis of an open-label, randomised, multicentre, phase 3 trial. Lancet Respir Med. 2022;10(1):72-82. doi:10.1016/S2213-2600(21)00166-1
15. Douillard JY, Ostoros G, Cobo M, et al. First-line gefitinib in Caucasian EGFR mutation-positive NSCLC patients: a phase-IV, open-label, single-arm study. Br J Cancer. 2014;110(1):55-62. doi:10.1038/bjc.2013.721
16. Mok TS, Wu YL, Thongprasert S, et al. Gefitinib or carboplatin–paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361:947-957. doi:10.1056/NEJMoa0810699
17. Soria JC, Ohe Y, Vansteenkiste J, et al; FLAURA Investigators. Osimertinib in untreated EGFR-mutated advanced non–small-cell lung cancer. N Engl J Med. 2018;378(2):113-125. doi:10.1056/NEJMoa1713137
18. Planchard D, Jänne PA, Cheng Y, et al; FLAURA2 Investigators. Osimertinib with or without chemotherapy in EGFR-mutated advanced NSCLC. N Engl J Med. 2023;389(21):1935-1948. doi:10.1056/NEJMoa2306434
19. Mok TS, Wu YL, Ahn MJ, et al. Osimertinib or platinum–pemetrexed in EGFR T790M–positive lung cancer. N Engl J Med. 2017;376(7):629-640. doi:10.1056/NEJMoa1612674
20. Goldberg SB, Pulla MP, Lisberg AE, et al. 123TiP: TROPION-Lung14: a phase III study of osimertinib ± datopotamab deruxtecan (Dato-DXd) as first-line (1L) treatment for patients with EGFR-mutated locally advanced or metastatic (LA/M) non-small cell lung cancer (NSCLC). J Thorac Oncol. 2025;20(suppl 1):S86-S87. doi:10.1015/S1556-0864(25)00319-3